You have successfully logged out.
August 8, 2024
B. Braun Issues Voluntary Nationwide Recall of 0.9% Sodium Chloride for Injection USP 1000 mL in E3 Containers Due to the Potential for Particulate Matter and Leakage
BETHLEHEM, PA - August 8, 2024 – B. Braun Medical Inc. (B. Braun), is voluntarily recalling two (2) lots of 0.9% Sodium Chloride for Injection USP 1000 mL in E3 containers within the United States to the consumer level. The voluntary recall has been initiated due to the potential for particulate matter and fluid leakage of the respective containers.
The affected batches were inadvertently released to the market prior to the completion of the required acceptance activities for embedded particulate matter which may result in leakage. To date, there have been no customer complaints received and there have been no reports of serious injury or death associated with this issue.
Risk Statement: There is a reasonable probability of embolic phenomena such as stroke or ischemia/infarct to other organs and possible infection if these particulates are not sterile that could lead to permanent damage or impairment of body function which could be life-threatening.
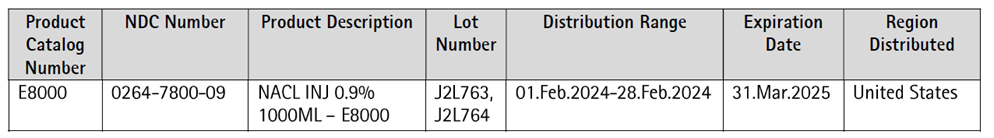
This intravenous solution is indicated for use in adults and pediatric patients as a source of electrolytes and water for hydration. 0.9% Sodium Chloride for Injection USP in E3 is indicated for extracellular fluid replacement, treatment of metabolic alkalosis in the presence of fluid loss and mild sodium depletion. 0.9% Sodium Chloride Injection USP is also indicated for use as a priming solution in hemodialysis procedures and may be used to initiate and terminate blood transfusions without hemolyzing red blood cells. Sodium Chloride Injection USP is also indicated as a pharmaceutic aid and diluent for the infusion of compatible drug additives. Product was distributed Nationwide within the United States to domestic distributors.
B. Braun has notified its distributors and customers by an official recall notice sent via certified registered mail and has arranged for return of all recalled products. Facilities and distributors that have product which is being recalled should discontinue use immediately and contact the B. Braun Medical Inc. Customer Support Department at 800-227-2862 Monday through Friday, 8 a.m. – 6 p.m. EST to arrange for product return.
Adverse reactions or quality problems experienced with this product, or questions about this recall may be reported to B. Braun’s Postmarket Surveillance Department by calling 1-833-425-1464.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
About B. Braun
B. Braun Medical Inc. (B. Braun) is a leader in smart infusion therapy and safe and effective pharmacy products, patient and provider safety, and sustainable health solutions. Our purpose is to help providers constantly improve patient satisfaction and outcomes. With products and services created to help healthcare professionals focus on what matters most—their patients—we’re uniquely positioned to help health systems succeed now and in the future. B. Braun is headquartered in Bethlehem, Pennsylvania and is part of the B. Braun Group of Companies in the U.S., which includes B. Braun Interventional Systems, Aesculap® and CAPS®. The company employs 8,500 people at over 30 locations across North America. Globally, the B. Braun Group of Companies employs more than 64,000 employees in 64 countries. Guided by its Sharing Expertise® philosophy, B. Braun continuously exchanges knowledge with customers, partners and clinicians to address the critical issues of improving care and lowering costs. To learn more about B. Braun Medical, explore our website.
