You have successfully logged out.
NRFit® Connectors
Reshaping safety for neuraxial and regional anesthesia
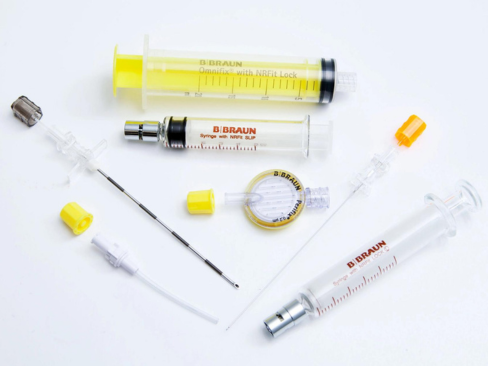
NRFit Connectors are built to avoid the risk of misconnections, helping to improve patient safety. With over 3,000 successful NRFit Connector conversions globally,1 B. Braun partners with facilities to help manage all aspects of the conversion, from clinical education to inventory logistics.
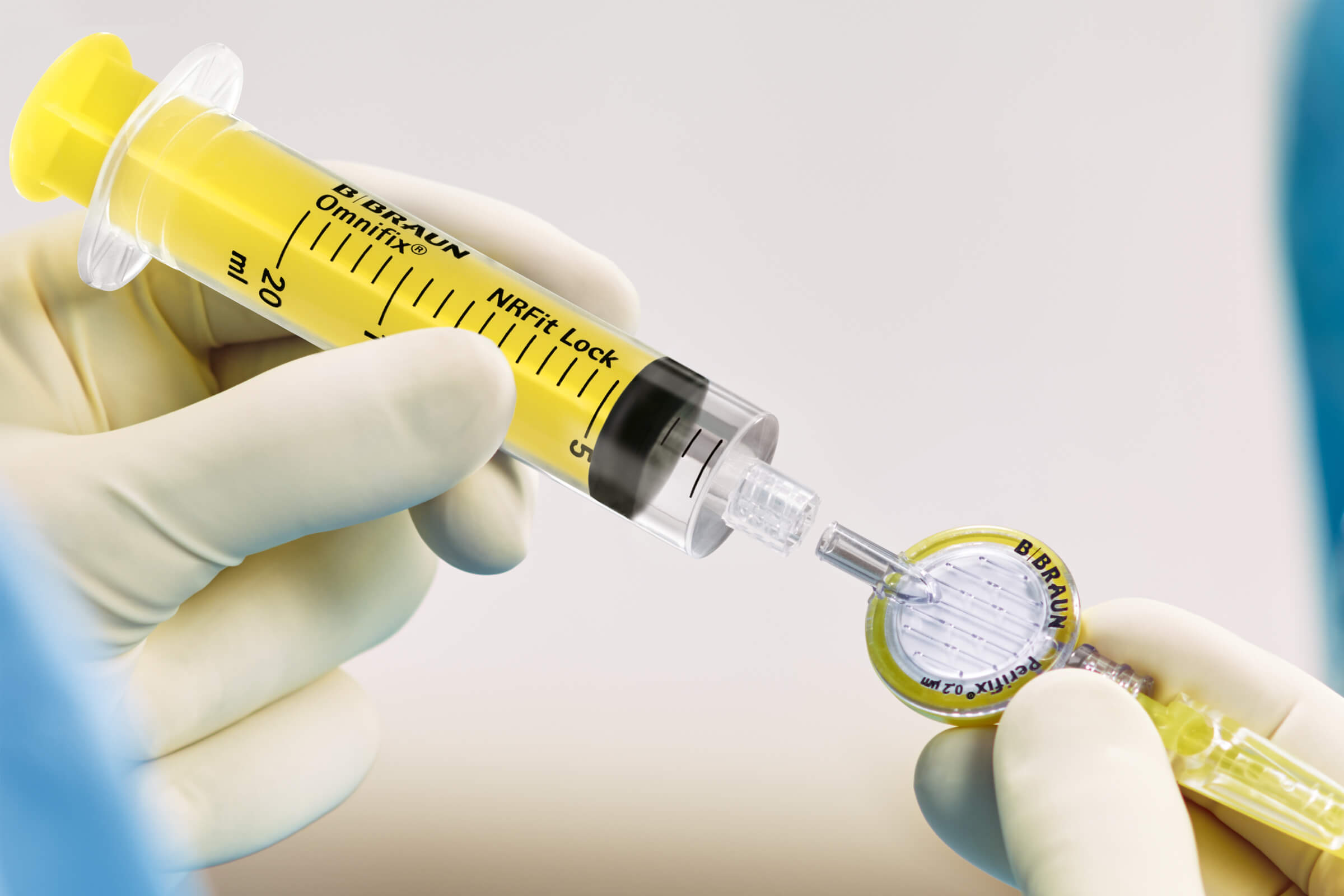
Addressing misconnections in regional anesthesia
For years, the main small-bore connector in the medical device field has been the Luer connector, which has been used in numerous applications, such as vascular, enteral, respiratory, epidural and intrathecal. The consequence of this has been that devices that were never intended to be connected can, in some circumstances, allow unintended fluids or gases to be delivered through the wrong route of administration.2, 3, 4
Promoting patient safety
B. Braun works with the International Organization for Standardization (ISO) to help develop standards for small-bore connectors that focus on neuraxial and regional anesthesia applications, aiming to prevent unintended fluid or gas delivery. The NRFit Connector mitigates cross-connections which helps improve patient safety. We are also proud to be a member of the Global Engineered Device Supplier Association (GEDSA), a nonprofit trade association dedicated to advancing patient safety.
References
NRFit® is a trademark of GEDSA and is used with their permission
- B. Braun Data on File
- GEDSA. Frequently Asked Questions. Retrieved from http://stayconnected.org/frequently-asked-questions.
- Tran, M. (2009, November 18). NHS Trust Prosecuted over New Mother's Death from Wrong Drug. The Guardian. Retrieved from https://www.theguardian.com/society/2009/nov/18/nhs-cabrera-wrong-drug-childbirth.
- Anger as Fatal Jab Doctor Freed. (2003, September 23). BBC News. Retrieved from http://news.bbc.co.uk/2/hi/health/3133076.stm.